✓ Solved: Calculate the pH after 0.10 mole of NaOH is added to 1.00 L of the solution in Exercise 35,...

Mole Review 1.) Calculate the number of moles in 60.4L of O2. 2.) How many moles are there in 63.2g of Cl2? 1 mol O2 60.4L O2 = 2.7 mol O2 22.4L
![P vs V graph is plotted for 1 mole of hypothetical gas. Range of a/b for this gas in (atm-L/mole). [R = 0.08 "atm-litre"//-"mle"-K] P vs V graph is plotted for 1 mole of hypothetical gas. Range of a/b for this gas in (atm-L/mole). [R = 0.08 "atm-litre"//-"mle"-K]](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/17243242_web.png)
P vs V graph is plotted for 1 mole of hypothetical gas. Range of a/b for this gas in (atm-L/mole). [R = 0.08 "atm-litre"//-"mle"-K]
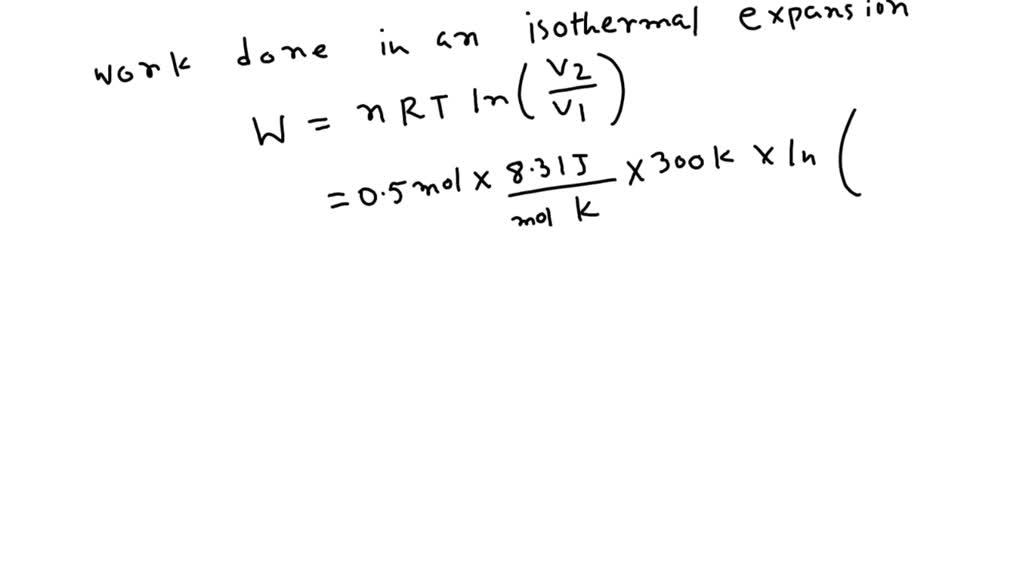








![Solved R=0.082 atm-L/mole-K R=8.314 J/mole-K 1. [20 pts] | Chegg.com Solved R=0.082 atm-L/mole-K R=8.314 J/mole-K 1. [20 pts] | Chegg.com](https://media.cheggcdn.com/media/a86/a86159ad-1549-4942-b7ad-79d47e1744ab/phpGTjsCE)